Synthesis of Lidocaine:
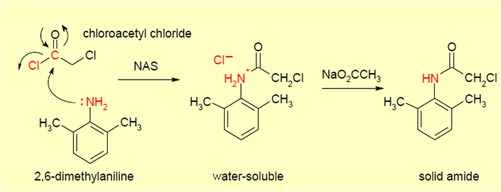
Raw Materials: 2,6-dimethylaniline and Chloroacetyl Chloride
Step 1
Nucleophilic Acyl Subsitution reaction (NAS):
- A substitution reaction in which nucleophiles - a compound that donates an electron pair during a reaction to form a chemical bond - reacts with an acyl - a compound with a functional group derived from a carboxylic acid - to result in a compound containing a carbonyl functional group. In this case, the nucleophile is 2,6-dimethylaniline as it donates an electron pair from the NH2 to replace Cl- and bond with the acyl, chloroacetyl chloride, through a NAS reaction to form a water soluble compound containing a carbonyl group called α-chloro-2,6-dimethylacetanilide.
- Sodium acetate is then added to the water soluble compound to form solid amide + CH3CO2H + NaCl. The two by products are not shown.
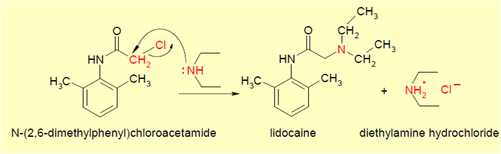
Step 2
SN2 reaction (Bimolecular Nucleophilic Substitution):
- SN2 reaction is a type of NAS reaction where the nucleophile, diethyl amine in this case, bonds after donating an electron pair.
- Lidocaine + diethylamine hydrochloride is formed.